If you are trying to understand what is inside a battery, the answer comes down to a simple but clever system. A battery stores energy in chemical form and converts it into electricity when connected to a device. Thinking about the battery parts involved can make this easier to grasp.
Although batteries power everything from small gadgets to large energy systems, they all rely on the same core structure and process. In other words, the components inside a battery follow similar principles across different chemistries.
Main Components Inside a Battery
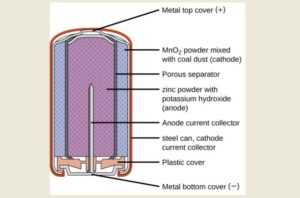
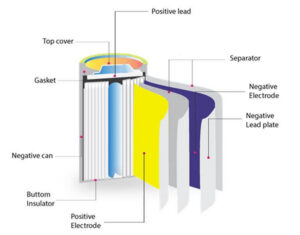
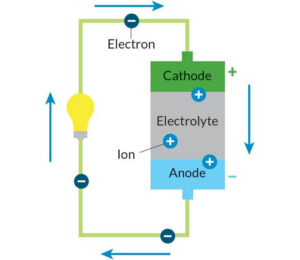
Every battery contains four essential parts—often called battery components—that work together in a continuous cycle. The anode acts as the negative electrode and releases electrons during use. The cathode serves as the positive electrode and receives those electrons after they pass through a connected device. Between them sits the electrolyte, which enables ions to move internally and keep the reaction balanced. A separator keeps the anode and cathode apart while still allowing this movement to happen safely. Each battery component plays a distinct role in this electrochemical process.
What Materials Are Used Inside a Battery?
The materials inside a battery vary depending on its type and purpose. Alkaline batteries commonly use zinc and manganese dioxide, while lithium-ion batteries rely on lithium compounds combined with carbon materials to achieve higher energy density. Lead-acid batteries, often used in vehicles, use lead plates and sulphuric acid.
These material choices directly affect performance, lifespan and environmental impact, as well as how batteries function in real-world applications. Each battery component is selected to balance performance, cost, safety and sustainability.
How Does a Battery Work?
When a battery is connected to a device, a chemical reaction begins. Electrons flow from the anode through the external circuit, powering the device, before returning to the cathode. At the same time, ions travel through the electrolyte inside a battery to maintain balance within the system.
This movement continues until the stored chemical energy is used up.
Why Do Batteries Lose Power Over Time?
One area where many articles fall short is explaining degradation. Over time, the chemical reactions inside a battery become less efficient. Materials break down, internal resistance increases and the battery can no longer hold or deliver the same amount of energy.
This is why rechargeable batteries gradually lose capacity and need replacing.
Why Understanding What Is Inside a Battery Matters
Knowing what is inside a battery helps you make better decisions when choosing batteries, understanding performance and recognising early signs of wear. Knowing the key battery parts also helps when comparing models or technologies.
It also highlights the importance of safe handling and responsible disposal.
How Does a Battery Produce Electricity?
A battery produces electricity by releasing electrons from the anode, which flow through a device to the cathode, while ions move through the electrolyte to maintain balance.
Why Do Batteries Degrade?
Batteries degrade because the internal chemical reactions become less efficient over time, causing a loss of capacity and performance.
Battery Safety and Recycling
Batteries contain materials that should not be thrown away carelessly. Improper disposal can lead to environmental damage and safety risks.
Recycling batteries ensures that valuable materials are recovered and harmful substances are handled correctly.