Easy Guide to Battery Science
Understanding how batteries work helps you see the science powering everyday devices and why proper disposal matters. Batteries store energy and convert it into electricity, powering everything from remote controls to laptops and electric vehicles.
Safe recycling of used batteries is essential because these cells contain valuable materials like lithium, nickel, cobalt, and lead that can be reused instead of ending up in landfill.
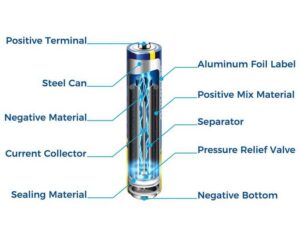
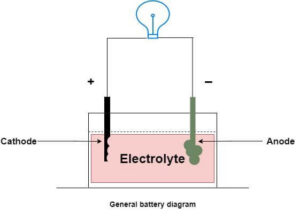
Most battery cells are electrochemical. Within each cell, chemical reactions create an electric current. These reactions occur between a negative terminal (anode) and a positive terminal (cathode) separated by an electrolyte that allows ions to move while electrons flow through an external circuit to power devices.
A battery is made up of anode, cathode, and electrolyte. When a device is connected, chemical reactions cause electrons to travel from the anode to the cathode, producing usable electricity. The exact chemistry depends on the type of battery, with common kinds including alkaline, lithium‑ion, nickel–metal hydride, and lead‑acid cells. Each type has different energy densities, applications, and recycling considerations. Spent cells should never be thrown in household waste; instead, follow appropriate recycling paths.
The Science Behind How Batteries Work
At its core, how do batteries work comes down to electrochemical energy. Inside a battery, oxidation and reduction reactions occur. The anode undergoes oxidation, meaning it loses electrons, while the cathode undergoes reduction, meaning it gains electrons. These electron movements produce an electric current when the battery is connected to a device. Rechargeable cells reverse this process during charging, restoring chemical potential.
Different battery chemistries influence performance and lifespan. Primary (non‑rechargeable) cells, such as alkaline cells, rely on chemical changes that are not easily reversible. Secondary (rechargeable) types, like lithium‑ion, can undergo reversible reactions that allow repeated charging cycles.
Battery Types
Battery designs vary widely in how they are built and where they are used. Primary cells, such as alkaline or zinc–carbon, are suited for low‑drain devices like toys, remotes, and flashlights. Rechargeable chemistries like lithium‑ion are common in modern technology, from mobile devices to electric cars, because they offer high energy density and the ability to be recharged many times. Nickel–metal hydride (NiMH) cells are also rechargeable and often found in hybrid vehicles and high‑drain consumer electronics.
Proper disposal of all these types is essential for environmental protection and resource recovery. UK‑based battery recycling services handle a wide range of battery types, from small portable cells to large automotive and industrial units, making sure they are processed safely and materials are recovered for reuse rather than polluting the environment.
Why Battery Power Fades
A battery loses power over time as the chemical reactants inside it get used up or degrade. In rechargeable cells, active materials can deteriorate after many charge cycles, reducing capacity. Environmental conditions such as extreme heat or deep discharge can speed up this degradation, shortening battery life. Understanding this helps users improve battery usage habits and prolong device runtime.
Battery Recycling in the UK
Recycling battery waste is not just about disposal; it’s about recovering valuable materials and preventing environmental harm. UK recycling systems collect used cells and transport them to licensed facilities where they are sorted and processed based on chemistry. Materials such as lithium, nickel, cobalt, lead, and plastics are separated, purified, and reused to manufacture new products, reducing the need for virgin raw materials. This helps support a circular economy.
Different sectors have specialised recycling needs. For example, EV battery recycling requires secure packing, safe transport, and careful dismantling because large lithium‑ion packs can pose fire and chemical hazards. Advanced recycling techniques recover metals like lithium and cobalt for reuse, reducing environmental impact. Similarly, automotive, portable, and e‑mobility battery packs are processed in ways that maximise material recovery while complying with strict safety regulations.
Responsible recycling also prevents toxic substances like lead, cadmium, and corrosive electrolytes from contaminating soil and water.
Practical Tips for Longer Battery Life
To extend battery life, avoid extreme temperatures, only partly discharge rechargeable batteries before recharging, and use chargers designed for specific chemistries. Remove cells from devices that won’t be used for long periods to prevent leakage or degradation. You can also find detailed guidance on battery maintenance tips on your site to help users improve battery performance and lifespan.